Rare respiratory diseases have long existed in the margins: too uncommon to attract sustained investment, too complex to solve in isolation, and too often invisible to the systems designed to support patients.
Fortunately, that is beginning to change. The convergence of genomics, improved diagnostics and a growing rare disease research infrastructure means we are genuinely close to transforming outcomes for patients who have waited far too long. New approaches to clinical trial design are making it possible to generate meaningful evidence in small populations. Regulatory frameworks are evolving to better reflect the realities of rare disease drug and novel therapeutic development. Innovative funding and partnership models is opening new pathways to bring therapies to the patients who need them.
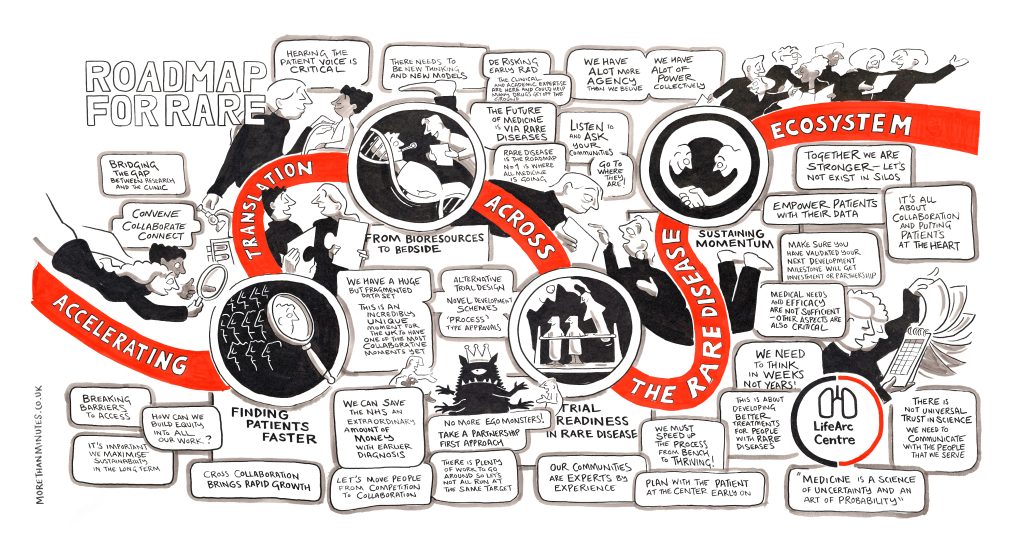
On 26 March, 2026, we brought together one hundred rare disease experts and advocates at Roadmap for Rare: Accelerating Translation Across the Rare Disease Ecosystem to ask whether we can move fast enough, and together enough, to make real progress happen.
Session I: Finding Patients Faster: Digital Innovation for Identification and Engagement

Rare respiratory diseases affect millions, yet the majority remain undiagnosed and invisible to the systems designed to serve them. Our first session at Roadmap for Rare asked a fundamental question on the road to treatment: how do we find the missing patients at scale and pace? Through the lens of health equity, NHS data infrastructure and cross-disease learning, our speakers explored the tools and partnerships transforming patient identification from a clinical challenge into a solvable problem. Because before we can act, we must first find.
Speakers
Priti Kenia, Consultant Paediatric Respiratory Medicine, Birmingham Women’s and Children’s Hospital NHS Trust
Louise Oni, Professor of Paediatric Nephrology, UCL & University of Liverpool
Mel Ivarsson, Chief Executive Officer of the new Health Data Research Service (HDRS)
Becca Cosgriff, LifeArc Scientific Director, Data Partnerships & Portfolio Strategy
Panel discussion chaired by Dr Harriet Holme, Exec Chair and Founder of PCD Research
Session II: From Bioresources to Bedside: Accelerating Discovery and Development

The path from scientific discovery to patient benefit is rarely straight, and in rare disease, it is rarely funded. The translational middle ground between early-stage research and clinical development remains the most difficult stretch of the journey: too advanced for basic science funding, too early for industry investment. This session explored how bioresources, connected patient data, and smarter preclinical funding models can bridge that gap. How they can build the evidence base needed to transform academic innovation into investable, trial-ready programmes? Because the science is often there. What is missing is a clear path to patients.
Speakers
Jason Mellad, Scientist Entrepreneur, Stealth Biotech start-up
Matthew Wood, Professor at University of Oxford, Director/CSO of the Oxford-Harrington Rare Disease Centre
Volker Straub, Director of the John Walton Muscular Dystrophy Research Centre, LifeArc Centre for Acceleration of Rare Disease Trials lead, University of Newcastle
Panel discussion chaired by Samantha Walker, Director of Research and Innovation, Asthma + Lung UK
Session III: Trial Readiness in Rare Disease: Design, Regulation and De-risking

Finding patients and generating the evidence needed to support new therapies are necessary steps, but not sufficient. To reach patients, therapies must navigate regulatory systems not designed with rare disease in mind. That is changing. In November 2025, the MHRA committed to a bold new framework for rare disease therapies, introducing adaptive trial designs, real-world evidence pathways and an investigative licensing model linking clinical trials and marketing authorisation in a single approval. Weeks later, the FDA unveiled its Plausible Mechanism Framework, allowing approval of individualised therapies for ultra-rare genetic diseases based on biological plausibility rather than large randomised trials.
This session explored what trial readiness means in this new landscape: how we design rare disease trials that regulators can approve, patients will join, and that generate evidence capable of changing clinical practice. The lessons are clear. Trials fail when patients aren’t at the table from the start, when outcomes aren’t defined with those living with the disease, and when the path to approval is navigated in isolation. The regulatory landscape is evolving. The opportunity now is to be ready for it.
Speakers
Jon Beaman, MHRA, Deputy Director Innovative Medicines
Janet Woodcock, former Principal Deputy Commissioner of the FDA, Director of Every Cure
Lorna Allen, CF Trust, Senior Involvement Manager
John Matthews, Chief Medical Officer, ReCode Therapeutics
Panel discussion chaired by Heather McKinnon, LifeArc Head of Chronic & Rare Respiratory Disease
Session IV: Sustaining Momentum: Partnerships, Investment and Long-term Impact in Rare Disease

Getting a therapy approved is not the finish line. Even landmark approvals leave patients behind through access barriers, eligibility gaps, or the limits of current science. Sustaining momentum means staying focused on all patients, not just those reached first.
Breakthroughs happen when science, patients, funding and infrastructure align. They stall when any one of those elements fails to hold. This session drew on the hard-won lessons of CF where decades of patient-driven investment delivered transformational therapies and asked what it takes to build that same infrastructure for other rare diseases, from world-class registries that have become engines for drug development, to financing models designed for the realities of rare disease translation, to the patient organisations that keep the pressure on long after the headlines have moved on. The roadmap does not end with a single approval: it ends when every patient who needs a therapy can access one and when the ecosystem is strong enough to keep finding the next path forward.
Speakers
Danny Gale, Director of National Registry of Rare Kidney Diseases (RaDaR), Professor of Nephrology, UCL
Pierre-Albert Colcomb, Ampleia, Chief Operating Officer
Emily Reuben, Founder and Chief Executive of Duchenne UK
Panel discussion chaired by Matthew Lumley, VP Clinical Research, Anlynam Pharmaceuticals
Photo credits: James Drury james-drury.com // Graphic minute by Jonny Glover morethanminutes.co.uk
